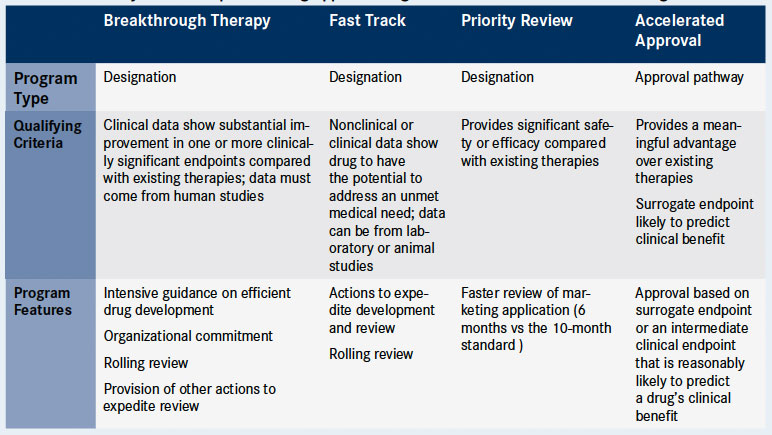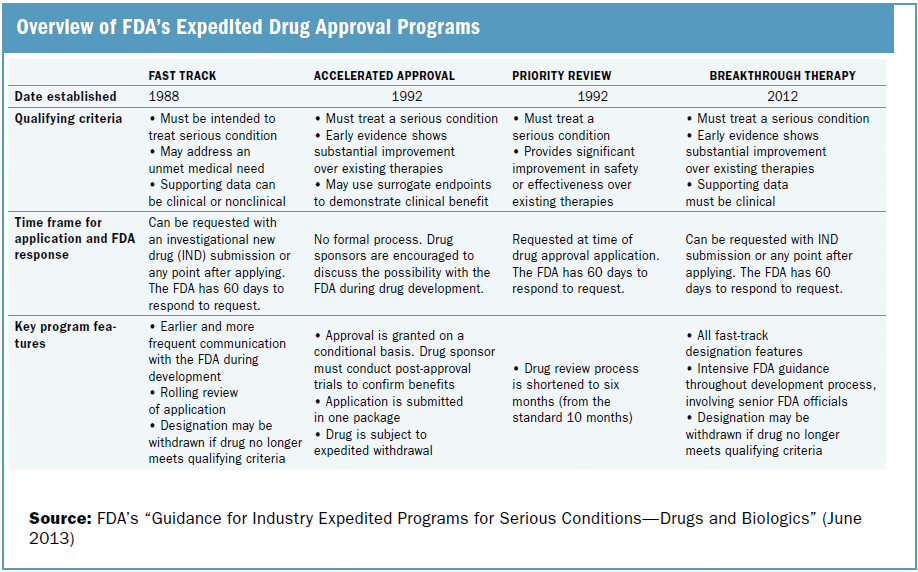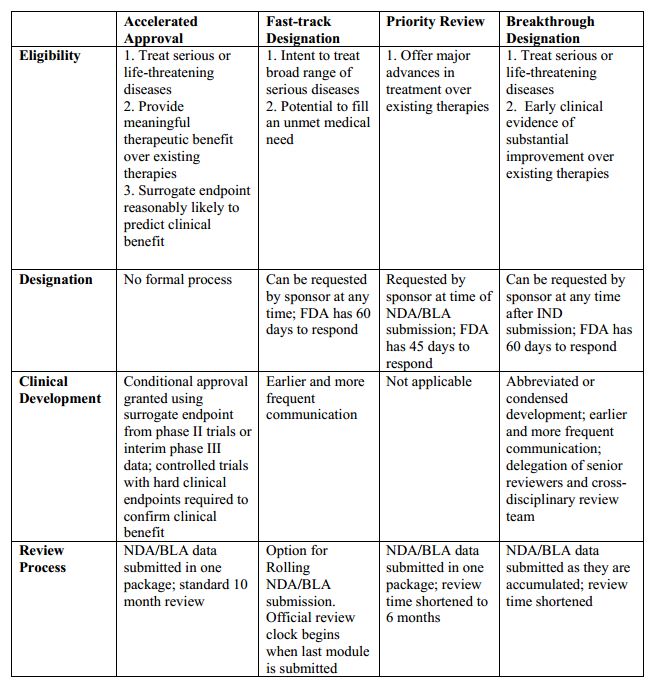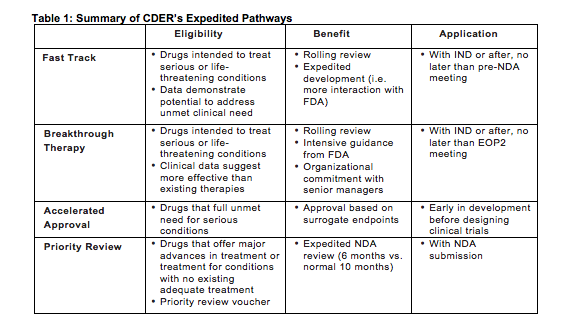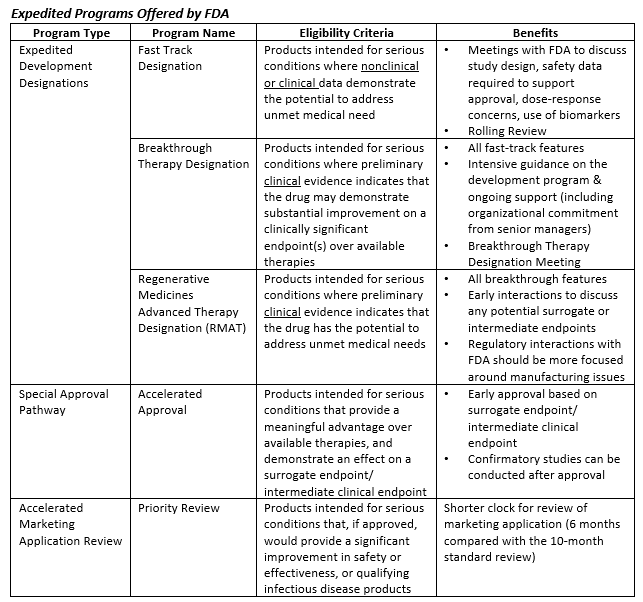
The Need for Speed in Drug Development: A Sponsor's Guide to FDA Expedited Programs | Halloran Consulting Group

More Ice in the Winter Time: FDA Breakthrough Therapy Designations – Great PR While Patients Suffer — Innovation Breakdown: How the FDA and Wall Street Cripple Medical Advances by Joseph V. Gulfo

Nassau EM - What is the difference between an Emergency Use Authorization (EUA) and the Food & Drug Administration's normal medication/vaccine approval process? Before any medication can be used or prescribed in

Frontiers | A Regulatory Risk-Based Approach to ATMP/CGT Development: Integrating Scientific Challenges With Current Regulatory Expectations
![PDF] Expediting drug development--the FDA's new "breakthrough therapy" designation. | Semantic Scholar PDF] Expediting drug development--the FDA's new "breakthrough therapy" designation. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7a27650b2c8093898b53aeb13b50266c96048cae/3-Table2-1.png)
PDF] Expediting drug development--the FDA's new "breakthrough therapy" designation. | Semantic Scholar