External Oxidant‐Free Regioselective Cross Dehydrogenative Coupling of 2‐Arylimidazoheterocycles and Azoles with H2 Evolution via Photoredox Catalysis - Chen - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

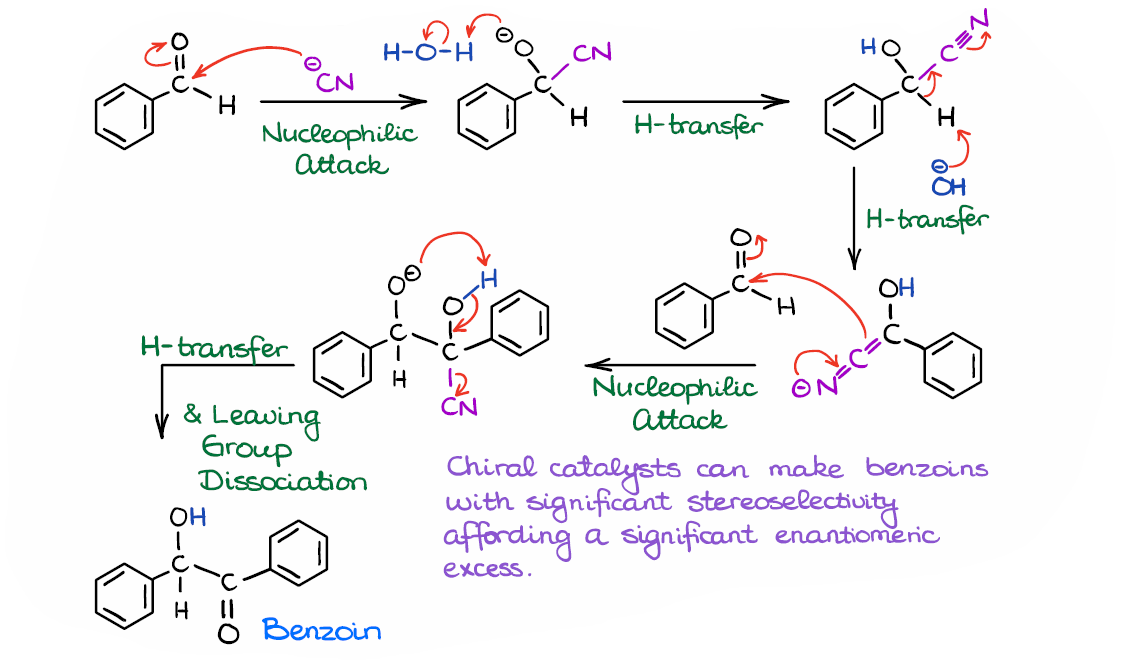
Thiamin‐Diphosphate‐Dependent Enzymes: New Aspects of Asymmetric CC Bond Formation - Pohl - 2002 - Chemistry – A European Journal - Wiley Online Library

Thiamin‐Diphosphate‐Dependent Enzymes: New Aspects of Asymmetric CC Bond Formation - Pohl - 2002 - Chemistry – A European Journal - Wiley Online Library

Regioselective Plasmon-Driven Decarboxylation of Mercaptobenzoic Acids Triggered by Distinct Reactive Oxygen Species | ACS Catalysis

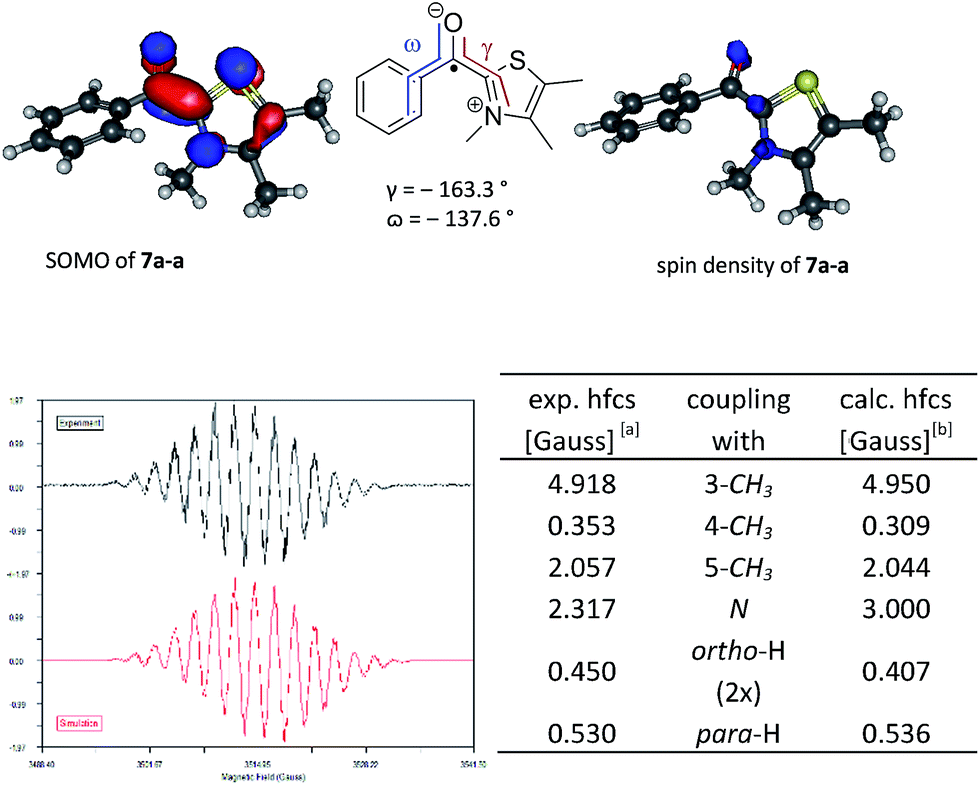
Can the Radical Channel Contribute to the Catalytic Cycle of N-Heterocyclic Carbene in Benzoin Condensation? | The Journal of Organic Chemistry

Small Molecule Activation Chemistry of Cu–Fe Heterobimetallic Complexes Toward CS2 and N2O | Inorganic Chemistry
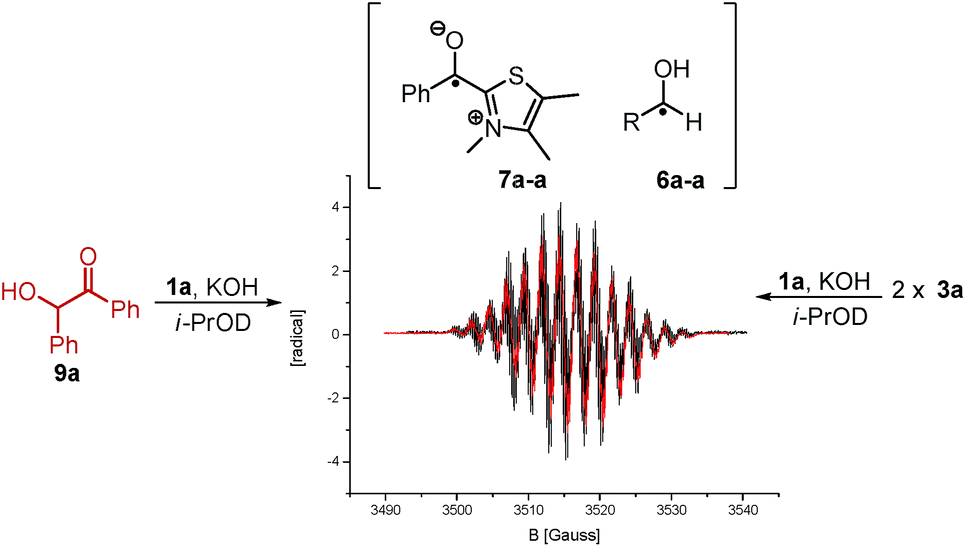
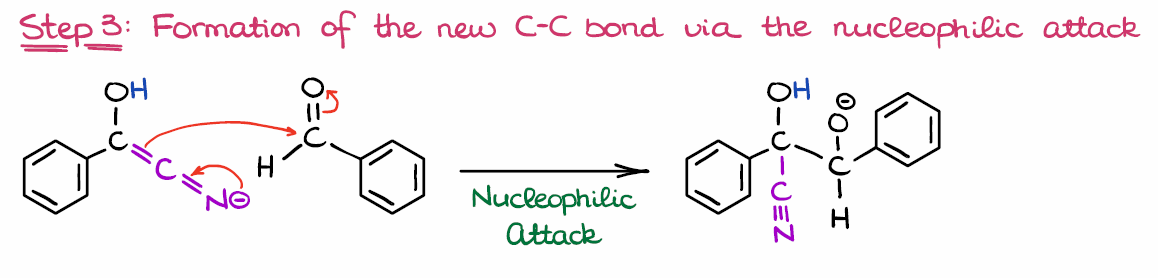
NHC-catalysed benzoin condensation – is it all down to the Breslow intermediate? - Chemical Science (RSC Publishing) DOI:10.1039/C5SC02186C

Can the Radical Channel Contribute to the Catalytic Cycle of N-Heterocyclic Carbene in Benzoin Condensation? | The Journal of Organic Chemistry

The Enzymology of Organic Transformations: A Survey of Name Reactions in Biological Systems. - Abstract - Europe PMC

Can the Radical Channel Contribute to the Catalytic Cycle of N-Heterocyclic Carbene in Benzoin Condensation? | The Journal of Organic Chemistry

Scheme of addition-condensation reaction of phenol-formaldehyde resins... | Download Scientific Diagram

Thiamin‐Diphosphate‐Dependent Enzymes: New Aspects of Asymmetric CC Bond Formation - Pohl - 2002 - Chemistry – A European Journal - Wiley Online Library

What Are the Radical Intermediates in Oxidative N-Heterocyclic Carbene Organocatalysis? | Journal of the American Chemical Society